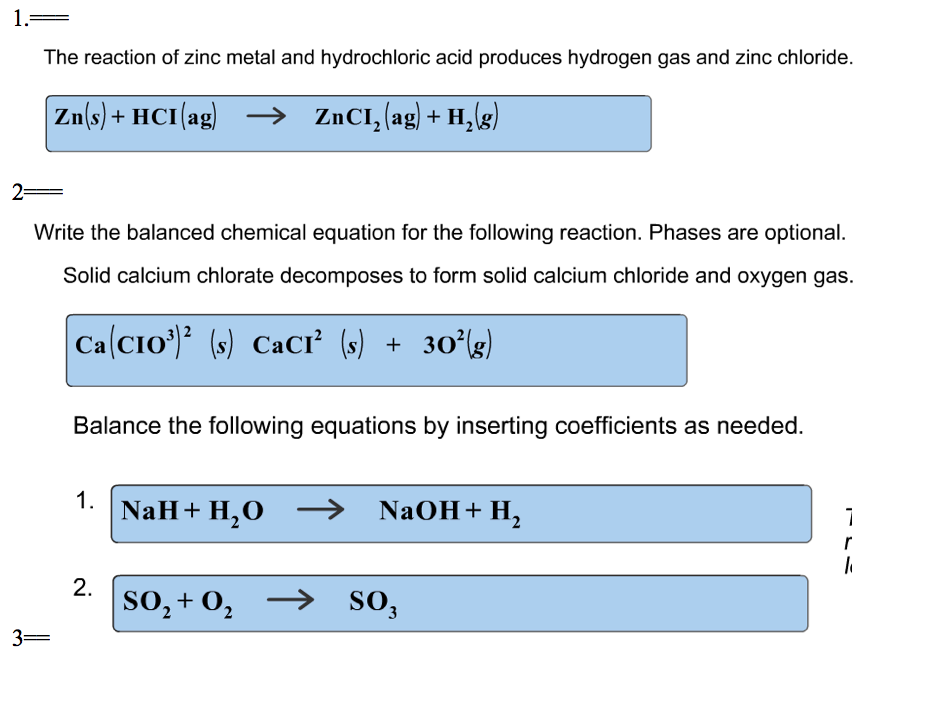
Unlocking the Potential of Hydrochloric Acid in Metal Processing and pH Regulation: It's a HCl of a Job – Alliance Chemical

The given diagrams show the reactions of three metals with dilute hydrochloric acid.What are metals P, Q and R ?P Q R

Emergency Information Panel - Hazchem Sign (EIP) Hydrochloric Acid - Company Name - Hartac Australia

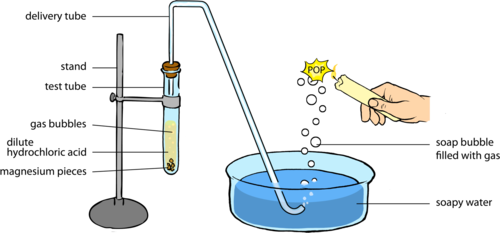
Question Video: Identifying the Name of the Gas Produced When Magnesium Metal Reacts with Hydrochloric Acid | Nagwa

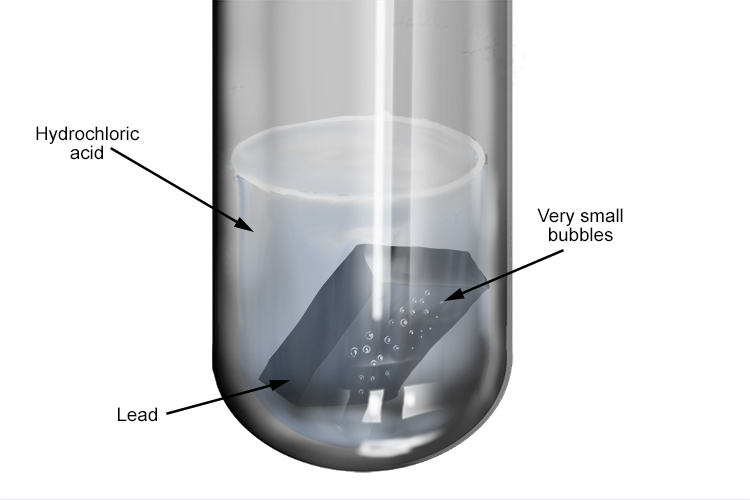
Experiment: The reaction of metals with dilute Hydrochloric acid - Free ZIMSEC & Cambridge Revision Notes

Fisher Chemical Hydrochloric Acid 34-37%, Trace Metal™, for Trace Metal Analysis, Fisher Chemical™ 1L Hydrochloric Acid | Fisher Scientific